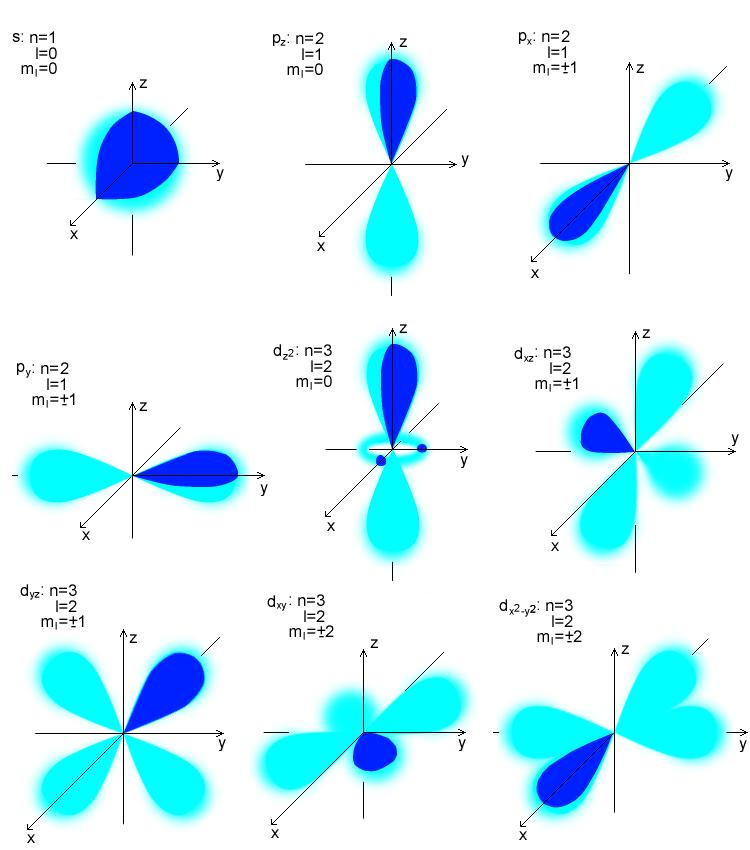
P orbitals in different shells are different sizes, just like s orbitals. As with s orbitals, p orbitals grow in size and energy as the primary quantum number rises (4p > 3p > 2p).The number of nodes is determined using the formula n – 2. The names 2px, 2py, and 2pz are given to the lobes because they are oriented along the x, y, or z-axis. The position of the lobes is the only distinction between the orbitals.The probability of finding an electron at the plane where the two lobes intersect is zero.īecause the three orbitals are identical in size, shape, and energy, they are degenerate orbitals. On either side of the plane that crosses the nucleus, each p orbital has lobes.The two lobes that make up each p orbital are situated on either side of the plane that crosses the nucleus.The p orbital can hold a maximum of six electrons due to the existence of three orbitals.The location of the p orbital node is in the nucleus’s center.The p orbitals have a dumbbell-like structure. 
The size progression is as follows: 1s, 2s, 3s,… The orbitals grow in size as the energy levels rise because the electrons are farther away from the nucleus. s-orbitalĪn s orbital has the nucleus of the atom at its center and is spherically symmetric around it, resembling a hollow ball made of relatively fluffy material. The s and p orbitals are the most utilized in organic and biological chemistry, making them the most significant of the four. The letters s, p, d, and f, which stand for sharp, diffuse, principal, and fundamental, are the symbols for the orbital shapes. The square of the orbital wave function represents the likelihood of finding an electron. The orbital wave function is the mathematical formula for the coordinates of an electron. The possibility of obtaining an electron close to the nucleus rises with a smaller orbital size. These orbitals fall into category based on their dimensions, shape, or inclination. Shapes of Atomic OrbitalĪccording to the quantum atomic model, an atom can have a very wide range of orbitals. The letters s, p, d, and f stand for the four different types of orbitals, each of which has a unique shape. The various atomic orbital shapes indicate which directions have the highest or lowest likelihood of having an electron around the nucleus. They provide a method for calculating the likelihood of discovering an electron in a specific area surrounding the atom’s nucleus.Ītomic orbitals have different shapes according to their type. Atomic orbitals are mathematical functions that describe how electrons (or electron pairs) behave in an atom.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
